Advantages
Advantages of sLH2 Cryotanks by SAG
Maximum Efficiency
Ranges of more than 1,000km for heavy duty truck applications due to high volumetric energy density of LH2 (compared to gaseous hydrogen storage solutions +40% at 700 bar & +60& at 350bar)
EU2021/535 compliant
Full compliance with the latest European hydrogen tank regulation for on-road safety and approval.
Proof of concept with 3rd generation tanks
Trucks with 3rd generation sLH2 storage tanks of SAG already under real-world conditions on public roads.
For FC & ICE
Useable for vehicles with fuel cell or ICE
Diesel-like Driving, Future-Proof Economics
Operational performance comparable to Diesel trucks, as well as cost efficient supply chain and total cost of ownership at scale
Scalable Cost Efficiency
Optimized for total cost of ownership and efficient logistics at fleet scale.
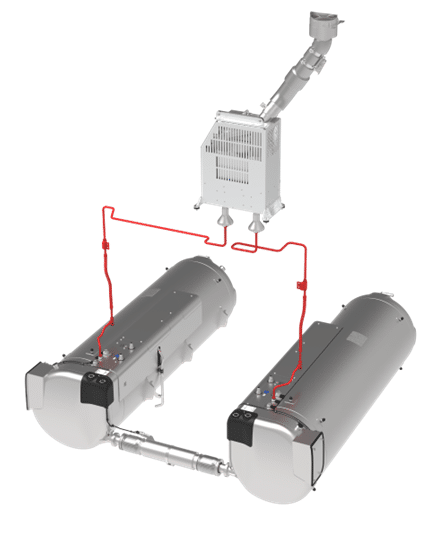
sLH2 Tanksysystem 3rd Generation
Technical details of sLH2 tank solution by SAG (addition)
- Double tank system based on single tank design
- Cryo Connection Line
- Boil Off Management System (BOMS)
- Reduced heat input
- Advanced tank pressure regulation
- Design adjusted for crash specifications
- Bonfire test and crash tests completed successfully
- EU535 compliant

SAG GROUP






